This trifecta has been prescribed since admission and likely before then; in this instance the trifecta refers to a proton pump inhibitor (PPI) plus frusemide plus digoxin. This trifecta significantly impacts magnesium status by:
- PPI inhibition of magnesium absorption,
- frusemide and digoxin both increasing magnesium excretion.
We do not know how much magnesium is not available to the body every day as a consequence of the impacts of the trifecta. Further we do not know how much magnesium is excreted as a direct consequence of the prescription of frusemide and digoxin in combination. We also don't know whether the body has an adaptive response to the sustained depletion of magnesium. Therefore we do not know how much magnesium needs to be added into the daily intake as either food or supplements to attain optimal intake.
Magnesium deficiency manifests as confusion, disorientation, personality changes, loss of appetite, depression, muscle cramps, tingling, numbness, hypertension, seizures, decreased absorption of thiamine, vitamin D and iodine, cardiac dysrhythmia and other signs and symptoms. Have you ever said “I think this person is losing weight because he/she has low magnesium?”
Magnesium is an intracellular ion therefore serum levels are unlikely to detect early depletion of status. Whilst cellular magnesium status is unknown, low magnesium levels typically indicate significant cellular depletion and intervention is recommended.
Men require 420 mg elemental magnesium per day and women require 320 mg elemental magnesium per day. A VERY common magnesium supplement in hospitals and residential care facilities, provides about 37.4 mg elemental magnesium per tab, and the typical dose is one tab per day. One does have to ask whether this dose is adequate.
Further, there are side effects from non-food sources that provide more than 350 mg elemental magnesium per day.
There is likely to be minimal benefit if magnesium supplements are administered whilst the trifecta is prescribed, especially given the presence of the PPI component. However, given calcium citrate supplements do not require gastric acidity for calcium absorption it is likely (speculatively) that magnesium citrate supplements may not require gastric acidity for absorption either. I am unaware of any evidence that this has been tested, so the speculation is based on first principles – something we in the nutrition field tend to depend upon when evidence is lacking.
Exacerbating the problem of magnesium deficiency is the broad variability in pathology ranges for acceptable levels, especially the cutoff level for lower acceptable limit. Personal observation has noted that various lower cutoff levels range between 0.6-0.85 mmol/L both clinically (pathology lab ranges) and in the research papers. Some researchers have questioned the current US reference range of 0.75-0.955 mmol/L for acceptable magnesium levels. The researchers further suggest that patients with risk factors for chronic disease such as glucose intolerance, inflammation, and elevated blood pressure, and who have magnesium levels within the range 0.75-0.85 mmol/L are currently deemed to have “normal” magnesium status, however may actually have chronic or subclinical deficiency also known as chronic latent magnesium deficiency. Further, the researchers propose an acceptable total serum magnesium concentration range to be 0.85-0.955 mmol/L.
It seems to me it has now become essential to check definitions of acceptable ranges when reading papers and reviewing pathology results in order to asses magnesium status. Further, for those whose magnesium levels may fall within the 0.6-0.85 mmol/L range, this variability also means that intervention is typically dependent upon the pathology lab’s defined acceptable range.
So, next time you see someone prescribed the trifecta will you integrate the following into your clinical assessment?
- check their magnesium status,
- request it to be checked if it is not available,
- check the pathology lab’s recommended range,
- comment accordingly especially if the defined lower limit is less than the proposed range of 0.85-0.955 mmol/L.
Given the increasing divergence between good food choices and poor diets, environmental impacts such as the trifecta can have significant negative impacts on magnesium status.

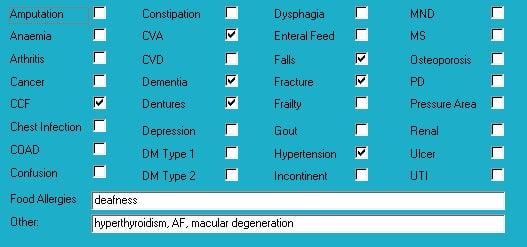
Medical History with Nutritional Aspect
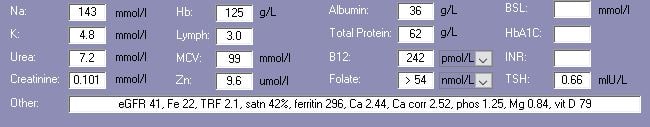
Biochemistry with Pharmaconutritional Consequences
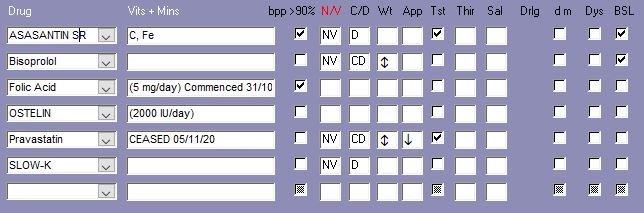
Medications That May Adversely Affect Nutritional Status
Comments – medication and nutrition impacts (direct and indirect) only
Recent relevant available biochemistry indicates marginal vitamin D – pravastatin may have negatively impacted vitamin D status. Useful vitamin D range is becoming quite controversial as most current pathology laboratory ranges are based on bone status impacts only and there is now an argument that has increasing traction that the lower limit of pathology ranges should be higher to facilitate other vitamin D-requiring physiological functions.
Regular aspirin consumption is a contributor to iron deficiency in the very elderly and decreases gastric vitamin C levels.
Assasantin SR and bisoprolol inhibit OCT2 (Organic Cation Transporter) a thiamine transporter to the kidney, therefore at increased risk of thiamine deficiency. Advisable to consider a thiamine intervention and administer thiamine intervention either one hour before or 2 hours after drug administration.
If a thiamine intervention is initiated then advisable to commence a magnesium intervention (to provide ~ 300 mg elemental magnesium/day) as adequate magnesium status is essential for activation of thiamine, vitamin D and iodine.
Folic acid has been prescribed for at least 1 year - evidence indicates excessive folic acid intake diminishes cognitive function. As folic acid levels are excessively high, I strongly recommend either reviewing necessity for its continued prescription and/or reducing frequency and dose of the intervention.
Statins such as pravastatin, significantly reduce plasma CoQ10 status - since pravastatin was only ceased 12 days ago, advisable to consider a short term (90-120 days) intervention to top up stores; CoQ10 is profoundly important in mitochondrial function.
Currently prescribed Slow-K however potassium levels well within acceptable range and no apparent cause for ongoing potassium depletion therefore advisable to review necessity for continued prescription of Slow-K.
Family advise Mrs AAA has been slowly losing weight over the last couple of years; the side effects profile of Asasantin SR, Bisoprolol, Slow-K, Pravastatin includes loss of weight.
Neomercazole and amiodarone were ceased 1 yr ago. Speculatively the sustained, high iodine intake from the amiodarone is likely to have impacted thyroid function and therefore neomercazole prescribed. As there is a likelihood of damage to the thyroid from the amiodarone, and whether that damage has healed, is unknown therefore advisable to check thyroid function and clarify current functional status.
What else would you include?

Yvonne Coleman is an accomplished dietitian with 30+ years of experience in aged care. Her mission is to make information concerning the interactions between medication and nutrition public and easy to access, having created the most comprehensive resource on the matter.
Her areas of competency include food science & nutrition, dietetics and health education. You can find out more about her work on LinkedIn, AusMed, the Enlightened Pharmacist podcast, and The FX Medicine Podcast.


