“Not to be crushed, chewed, broken or dissolved” and its many variations are commonly included in most medicines Product Information documents. Therefore, if a medicine is crushed, chewed, broken or dissolved to ensure safe administration then there is non-compliance with manufacturer’s instructions. Further, crushing, chewing, breaking or dissolving a medicine means alteration to some aspects of the medicine’s effects.
Commonly 20-60+% of residents within Aged Care facilities require texture-modified foods and fluids; safe administration of their medicines necessitates compliance with these texture modification requirements. This means there is often a direct conflict between manufacturer’s instructions and the Duty of Care in the safe administration of a person’s prescribed medicines.
In fact, all choking-related deaths in Residential Aged Care in Victoria are now investigated by the Coroner.
Some (not very many at all) pharmaceutical companies are now advising administration strategies in one or more of the following scenarios –
- reluctance to swallow whole tablets or capsules,
- impaired swallow reflex,
- enteral feeding.
The Impaired swallow reflex administration requirements for oral and enteral intake differ –
- oral intake – foods and fluids are modified to a defined safe swallowing consistency; mouthfeel and taste are both important,
- enteral intake – the administered formula is fluid and smooth; mouthfeel and taste are irrelevant however tube blockages and their prevention are significant issues.
A general recommendation is to always administer modified medicines with the same administration substance such as yoghurt, applesauce, etc. The use of a variety of administration substances can alter the availability and effectiveness of each modified medicine as there is potential risk for interactions between the administration substance(s) and the medicine(s) various ingredients.
The Dietitians and Speech Pathologists have collaborated to both create and establish a global standard for texture modification of foods and fluids with the International Dysphagia Diet Standardisation Initiative, https://iddsi.org/. This Standard has resulted in both a global standardized terminology and a global provision of uniform consistencies of texture modified foods and fluids. Ultimately this global Standard now means a person can change countries and still be given appropriately modified foods and fluids. The instigators and drivers of this global standard are to be applauded for their vision and ability to make it happen.
Now it is time for IDDSI-compliance to be integrated into the entire global healthcare system. This requires each Department in every healthcare-related organization to both review their direct and indirect contributions to a person’s formal and informal food and fluid intake, and then to develop safe administration strategies for each of the IDDSI-defined texture levels including administration via enteral feeding tubes. Inclusion of informal intake means most Departments are impacted.
- Direct intake includes Diet Therapy, Speech Pathology, Pharmacy, Radiology (imaging substances), Activities/Diversional Therapy, Food Services, etc.
- Indirect intake includes access to a water cooler, a clinician or associate offering a drink and/or something to eat whilst waiting for or during an intervention – for example a podiatrist offering a drink whilst feet are being inspected.
Until there are regulatory requirements for inclusion of IDDSI-compliant texture modification administration strategies in the Product Information documents, I suggest recording the decision processes in relation to altering the texture of a person’s prescribed medicine(s) in both your Notes and the medicine-recipient’s Notes, and to actively include the following –
- whether the supplying pharmacist and/or Accredited Pharmacist were contacted with regard to the conflict between GP prescription and Manufacturers administration recommendations as stated in the Product Information document;
- noting whether a directed response was sent to the GP (likely by a Pharmacist, Accredited Pharmacist, or Clinical Care Co-ordinator) advising that the Product Information states the prescribed formulation is not to be crushed, chewed, broken or dissolved and what action do they (the GP) recommend, and could they write that order in both the Notes and on the drug chart;
- whether there are alternative formulations for the prescribed medicine(s), and if available, the reason(s) they are not prescribed;
- whether the pharmaceutical company was contacted regarding administration of their product in this scenario, and their response (either written, or verbal as a Record of Conversation);
- whether your Medication Advisory Committee has developed guidelines for drug administration when texture modification is required, when it was last updated, and the audited level of compliance;
- whether the recommended administration substance be documented on the Drug Chart was agreed to.
Clinical Questions
What actions will you initiate as you review the nutritional implications of the prescribed medicines of those who require texture modified foods and fluids, will you –
- check that strategies are in place, and regularly audited, to ensure that all medication administration staff are familiar with, and comply with, the IDDSI and/or facility guides in relation to administering medicines to the prescribed level of safety for each person?
- check if the administration substance is documented on the Drug Chart and if not then check if there are guidelines in place, and frequent regular audits, to ensure consistency of use of approved administration substances only?
- write to the TGA (Therapeutic Goods Administration), FDA (Food & Drug Administraiton) and EMA (European Medicines Agency) and request that IDDSI-compliant texture modification strategies be included in the Product Information documents as a standard practice?
- initiate strategies for facility/organisation-wide integration of IDDSI-compliant strategies into all aspects of relevant healthcare service provision?
Conclusions
Not to be crushed, chewed, broken or dissolved has many implications for the safe administration and consumption of prescribed medicines when texture-modification is essential.
Disclaimer
The information in this article is provided to support Health Professionals. It is not an exhaustive protocol and Health Professionals are advised that adequate professional supervision be accessed to ensure that Duty of Care obligations with respect to safe administration of medicines is met for each consumer.

Medical History with Nutritional Aspect

Biochemistry with Nutritional Aspect

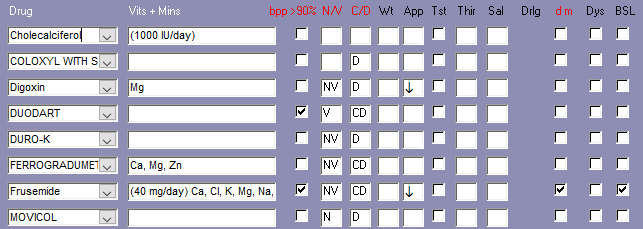
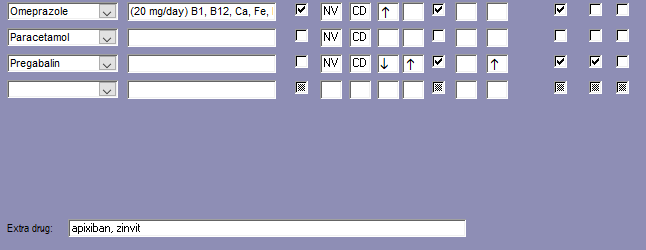
Medications That May Adversely Affect Nutritional Status
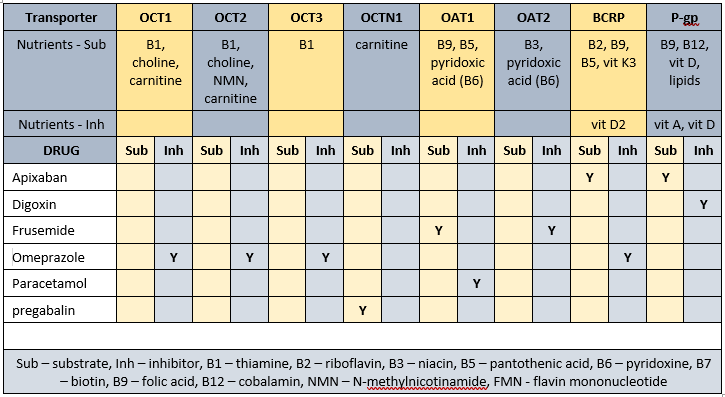
Transporter-mediated interactions and nutrients
Comments – medication and nutrition impacts (direct and indirect) only
Data summary
Biochemistry
Relatively recent biochemistry indicates -
- low Hb - associated with increased risk of falls, and poor appetite; currently prescribed omeprazole;
- low B12 – currently prescribed omeprazole therefore intervention recommended. There is disagreement between pathology ranges and research findings with regard to appropriate B12 levels - neuro-imaging research indicates a link between B12 status and consequent memory impairment; they also found increasing memory impairment as B12 levels dropped even whilst within currently defined acceptable ranges. The authors recommend B12 interventions once levels are less than 300 pmol/L;
- marginal vit D - currently prescribed an intervention therefore advisable to monitor current vitamin D management strategy;
- low Fe + satn - likely due to omeprazole effect. Advisable to recheck status and if still low then short term (90-120 days) intervention advisable however effectiveness of intervention is uncertain whilst omeprazole prescribed.
Glycaemia
Currently prescribed 1 medication that alters glycaemia.
Pharmaconutrition
Apixiban side effects include nausea, vomiting, constipation, diarrhoea, pain, chest pain, somnolence, hypokalaemia.
Chronic use of coloxyl + senna may promote excessive loss of water and electrolytes, especially potassium, and their regular monitoring recommended.
Digoxin increases urinary excretion of magnesium.
Frusemide increases urinary excretion of calcium, magnesium, potassium and sodium.
There is an argument that anti-epileptic drug treatments such as lyrica cause subnormal vitamin levels and suggest prophylactic supplementation to be an acceptable alternative to regular measurements and selective supplementation,
Ozmep decreases B12, vitamin C, magnesium, zinc and iron absorption, may decrease calcium absorption, and decreases thiamine availability.
Dietary levels of caffeine intake in conjunction with paracetamol inhibit antinocieception.
Mr ACN is prescribed the trifecta ie three drugs that decrease magnesium availability - being frusemide, ozmep and sigmaxin. Magnesium deficiency manifests as confusion, disorientation, personality changes, loss of appetite, depression, muscle cramps, tingling, numbness, hypertension, cardiac dysrhythmia, seizures. Magnesium is an intracellular ion therefore serum levels are unlikely to detect early depletion of status. Cellular magnesium status is unknown whilst magnesium levels within acceptable range however if magnesium levels are low then typically indicates significant cellular depletion and intervention recommended. Advisable to check magnesium levels and ensure > 0.8 mmol/L.
Bowel management
Regular aperient prescribed.
Oral + anal PRN interventions prescribed.
No Nurse Initiated interventions administered.
Mr ACN is prescribed 2 medicines for constipation management as well as 7 medications that include constipation as a side effect and 11 medications that include diarrhoea as a side effect.
Staff comments
Staff advise Mr ACN eats well.
Mr ACN is a charming slender man who had just finished eating his main course when I went to speak to him - he told me he eats well.
Mr ACN has steadily gained weight since admission ie for the last 2 years. It is unusual to observe such a sustained weight gain in a resident and therefore one has to question whether there is an underlying pathology.
Pharmaconutrition comments
Mr ACN’s diagnoses include chronic pain - nutritional factors that may be useful to consider in pain management include -
- vitamin D - current intervention may not be adequate to attain adequate range. Evidence indicates increasingly brittle pain control with decreasing vitamin D levels therefore advisable to monitor vitamin D levels on a regular basis; digoxin negatively impacts vitamin D status via inhibition of P-gp, and omeprazole negatively impacts vitamin D status via inhibition of BCRP;
- vitamin C - pain increases the reactive substances (formerly Reactive Oxygen Species) within cells. Vitamin C is important in quenching reactive substances and if there is insufficient vitamin C then cell status becomes compromised and the cells typically die which also causes pain. Advisable to consider a vitamin C intervention - the optimal intervention is 500 mg vitamin C/day (if more than 500 mg vitamin C administered at a time then the excess above 500 mg is not absorbed as the vitamin C transporters are overloaded). Vitamin C is not considered part of the pain management armament however it won't cause harm and evidence suggests it may confer benefit, especially since omeprazole negatively impacts vitamin C status by inhibiting its activation;
- evidence indicates relief of neuropathic pain by thiamine, pyridoxine and cobalamin separately, and in combination there was a synergistic benefit – these 3 nutrients are negatively impacted by several drug-transporter interactions;
- vitamin K - has been found to suppress the inflammatory cytokines and NF-kappaB and prevent oxidative, hypoxic, ischemic injury to oligodendrocytes and neurons – vitamin K deficiency therefore results in classic expression of the inflammatory response and consequently pain; omeprazole negatively impacts vitamin K3 status via inhibition of BCRP;
- low B12 exacerbates elevated TNF- α which is an inflammatory response marker; elevation of the inflammatory response can include a pain response and currently prescribed omeprazole therefore advisable to check B12 status. There is disagreement between pathology ranges and research findings with regard to appropriate B12 levels with neuro-imaging research showing a direct causal link between B12 status and memory impairment. Omeprazole negatively impacts B12 status directly by altering gastric pH and thus absorption, and also by inhibiting P-gp.
What else would you include?

Yvonne Coleman is an accomplished dietitian with 30+ years of experience in aged care. Her mission is to make information concerning the interactions between medication and nutrition public and easy to access, having created the most comprehensive resource on the matter.
Her areas of competency include food science & nutrition, dietetics and health education. You can find out more about her work on LinkedIn, AusMed, the Enlightened Pharmacist podcast, and The FX Medicine Podcast.


